General Information
Tums is an antacid made of sucrose and calcium carbonate, manufactured by GlaxoSmithKline in St. Louis, Missouri, USA. It is an over-the-counter drug and available at many retail stores, including drug stores, grocery stores and mass merchandisers. It provides relief from acid indigestion, heartburn, and indigestion and is considered a calcium supplement. Tums comes in chewable tablets that are taken orally. It is also available in a variety of different flavors such as peppermint and fruits such as "Assorted Fruit, Assorted berries, Assorted Mint,and Assorted Tropical Fruit."(TUMS® FAQ)
Chemical Information
scientific name
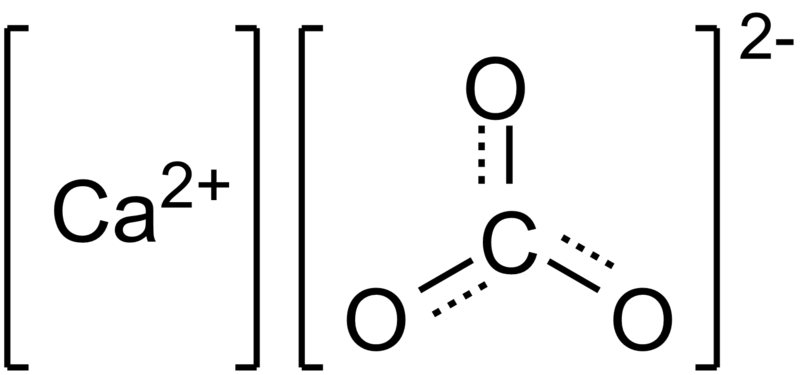
 Figure 2 Calcium Carbonate
Figure 2 Calcium Carbonate
The scientific name for Tums is calcium carbonate (KAL see um KAR boe nate) TUMS® tablets contain calcium carbonate USP. The calcium carbonate in TUMS® antacid is obtained from limestone via a chemical process which produces a high degree of purity. The calcium carbonate used in Tums is 99.5% pure and does not contain bone meal or oyster shell.(TUMS® FAQ,2014)
Chemical Formula
Calcium carbonate is a chemical compound with the chemical formula CaCO3. It is a common substance found in rocks in all parts of the world, and is the main component of shells and marine organisms, snails, pearls, and eggshells. Calcium carbonate is the active ingredient in agricultural lime, and is usually the principal cause of hard water. It is commonly used medicinally as a calcium supplement or as an antacid, but excessive consumption can be hazardous.("Calcium Carbonate.-Princeton University)
Physical Properties
Calcium carbonate (CaCO3) is a white solid (powder or crystal). It is odorless and tastes chalky or can have no taste at all. The molecular weight is approximately 100.09 with the density of 2.7-2.9. The melting/freezing temperature is 825 degrees Celsius. This material is slightly soluble in water, alcohol, and dilute acids . ("Material Safety Data Sheet," 2008, Section 9)
Chemical Properties
When calcium carbonate is exposed to acids such as hydrochloric acid, it chemically reacts by releasing carbon dioxide ("Calcium carbonate" pg. 1). Calcium carbonate is a non-corrosive material (Meister, 2006, pg. 6). The Ph is 8 to 9 ("Material Safety Data Sheet," 2008, Section 9). When heated, it reacts creating calcium oxide (CaO) and carbon dioxide gas (CO2). It also reacts vigorously with acids to release a froth of carbon dioxide bubbles. ("Calcium Carbonate." The Gale Encyclopedia of Science. Ed. K. Lee Lerner and Brenda Wilmoth Lerner. 3rd ed. Vol. 1. Detroit: Gale, 2004. 670-671. Gale Virtual Reference Library. Web. 4 Mar. 2014.)
Other Common Names
- Aragonite
- Agricultural limestone
- Ag stone
- Pulverized limestone
- Calcite
- Chalk
- Domolite
- Limestone